Photosensitizers and Applications
Renjith Thomas, T Pooventhiran
Renjith Thomas, Anila Skariah
A.A. Khairbek, M.I. Al-Zaben, A.Y.A. Alzahrani, R. Puchta, R. Thomas, Mechanistic and electronic structure insights into formate formation in uranium and related actinide complexes, Dalt. Trans. (2026). https://doi.org/10.1039/D6DT00462H
S.C. Sasi, S.A. Sunny, R. Sulay, R. Thomas, R. Krishnan, Exploring the Role of Hydrogen Bonding in Cyclodextrin─Naphthalenediimide Binding Orientation, J. Phys. Chem. B 130 (2026) 4234–4246. https://doi.org/10.1021/acs.jpcb.6c00517
R. Thomas, M.I. Al-Zaben, A.Y.A. Alzahrani, R. Puchta, A.A. Khairbek, A Comparative DFT Study of Pd- and Ni-Based Catalysts in the Narasaka–Heck/C(sp³)–H Activation Reaction, J. Comput. Chem. 47 (2026) e70361. https://doi.org/10.1002/jcc.70361
This work is dedicated to Prof. Dr. Peter R. Schreiner, who is celebrating his 60th birthday, in recognition of his remarkable scientific contributions and distinguished career.
M. Kattoor, I.V. Alabugin, R. Thomas, Electron Upconversion Enables C–P and C–S Bond Formation Under Mild Oxidative Conditions: A Theoretical Study, J. Comput. Chem. 47 (2026) e70319. https://doi.org/10.1002/jcc.70319
S.A. Sunny, R.F. Rajan, S. Thilakan, F. Thomas, T. Pooventhiran, A.Y.A. Alzahrani, R. Thomas, Investigating the Role of Noncovalent Interactions in Vadadustat’s Solubility and Stability With Selected Solvents, ChemistrySelect 11 (2026) e05824. https://doi.org/10.1002/slct.202505824
R. Sulay, S.A. Sunny, S. Bushramol, A.N. Arshana, A.Y.A. Alzahrani, R. Thomas, The Hydration Blueprint of Polylactic Acid: Computational Decoding of Noncovalent Interactions for Predictive Biodegradation, J. Comput. Chem. 47 (2026) e70322. https://doi.org/10.1002/jcc.70322
M. Mathew, R. Puchta, R. Thomas, Protonation-Induced Planarization and Aromaticity Enhancement in Diazahelicenes: A Route to Stronger Bases, J. Phys. Chem. A. (2026). https://doi.org/10.1021/ACS.JPCA.5C06393.
R. Thomas, A Systematic Computational Protocol for Deconstructing Non-Covalent Interactions: BerchNCI 1.0, AIDASCO Rev. 3 (2025) 28–40. https://doi.org/10.59783/AIRE.2025.84.
S.A. Sunny, T. Mathew, J.M. Thomas, A.Y.A. Alzahrani, R. Thomas, Dominant non-covalent forces in diphenhydramine-8-chlorotheophylline supramolecular assembly, Mod. Phys. Lett. B. 40 (2026). https://doi.org/10.1142/S0217984925502859.
R. Thomas, A.Y.A. Alzahrani, A.A. Khairbek, Halogen-Substituted Binuclear Copper Complexes as Efficient Catalysts for Azide–Alkyne Cycloaddition Reactions, Appl. Organomet. Chem. 39 (2025). https://doi.org/10.1002/aoc.70419.
A.A. Khairbek, A. Moulhim, H. Ibraheem, A.Y.A. Alzahrani, P. Thangaiyan, R. Thomas, Combining data-driven and quantum chemical approaches to evaluate Minnesota functionals (M06-2X, MN12-SX, and MN15) for the enthalpy of formation predictions, J. Mol. Model. 31 (2025). https://doi.org/10.1007/s00894-025-06583-6.
A.A. Khairbek, M.I. Al-Zaben, R. Thomas, Assessing the Catalytic Potential of Novel ADAP-M (M = Cu, Ag, Au) Catalysts in [3 + 2] Cycloaddition Reactions, J. Comput. Chem. 46 (2025). https://doi.org/10.1002/jcc.70194.
S. Masroor, M.S. Hussain, A. Kumar, U. Ishrat, S.A. Sunny, R. Thomas, M. Rizvi, M. Mustaqeem, M.E. Khan, Hybrid TiO2–polymer composites for corrosion protection: a combined gravimetric, DFT, and molecular docking approach, J. Mol. Liq. 438 (2025). https://doi.org/10.1016/j.molliq.2025.128773.
J.S. Al-Otaibi, M. Hossain, R. Thomas, Y.S. Mary, I. Habib, S. Armaković, S.J. Armaković, C. Van Alsenoy, Newly synthetized imidazole derivatives – structure, spectroscopy and reactivity investigated by combination of experimental and computational approaches, J. Mol. Liq. 439 (2025). https://doi.org/10.1016/j.molliq.2025.128771.
K.J. Rajimon, R. Almeer, P. Thangaiyan, R. Thomas, Structural, spectroscopic, and biological properties of an asymmetric Cu (II) Schiff-base complex: Synthesis, DNA/BSA interactions, and antimicrobial potential with experimental and computational insights, Polyhedron. 265 (2025) 117277. https://doi.org/10.1016/j.poly.2024.117277
K.J. Rajimon, A.Y.A. Alzahrani, P. Thangaiyan, R. Thomas, Multifaceted potency of green synthesized pyridine derived Schiff base: A comprehensive study of antimicrobial, antioxidant, anti-inflammatory, cytotoxic, and Enzymatic properties with density Functional Theory and bioinformatics insights, Biochem. Biophys. Res. Commun. 792 (2025). https://doi.org/10.1016/j.bbrc.2025.152959.
K.J. Rajimon, B.K. Sarojini, R. Thomas, Investigating the intrinsic electronic, structural properties and diverse bioactivities of mechanochemically crafted halogenated pyridine derivative: A combined experimental and computational study, Biochem. Biophys. Res. Commun. 783 (2025) 152606. https://doi.org/10.1016/j.bbrc.2025.152606.
K.J. Rajimon, H.A. El-Serehy, D.S. Rajendran Nair, N. Kannan, A. Jayashree, R. Thomas, A Comprehensive Journey of a Vanillic Aldehyde-Chloroaniline Schiff Base from Solvent-Free Synthesis to Electronic Structure, In Silico and In Vitro Biological Analysis, J. Comput. Biophys. Chem. 24 (2025) 189 – 213. https://doi.org/10.1142/S2737416524500571.
R. Karthika, S. Nandhini, S. Suriya, T. Pooventhiran, A.Y.A. Alzahrani, R. Thomas, Synthesis of Vanillin derivative Schiff’s bases from threonine, and valine: Structural study and multiscale computational modeling, Biochem. Biophys. Res. Commun. 781 (2025) 152501. https://doi.org/10.1016/j.bbrc.2025.152501.
W. Daoudi, K. karrouchi, M. Alqarni, I.A. Naguib, B. Tüzün, R. Thomas, E. Natarajan, S. Arshad, A. Oussaid, A. El Aatiaoui, Synthesis, Single Crystal X-Ray Diffraction Analysis, Hirshfeld Surface, and Molecular Docking Study of 7-Methyl-2-phenylimidazo[1,2-a]Pyridine-3-carbaldehyde, ChemistrySelect. 10 (2025). https://doi.org/10.1002/slct.202504019.

A Systematic Computational Protocol for Deconstructing Non-Covalent Interactions: BerchNCI 1.0
Non-covalent interactions constitute the fundamental organizing principles of supramolecular assemblies; however, the accurate modeling of these subtle, dispersion-driven forces remains a formidable challenge in theoretical chemistry. In this work, we formally propose the Berchmans Protocol for Modelling Non-Covalent Interactions 1.0 (BerchNCI 1.0), a comprehensive, hierarchical computational workflow designed to decipher the electronic anatomy of NCIs with benchmark precision.
View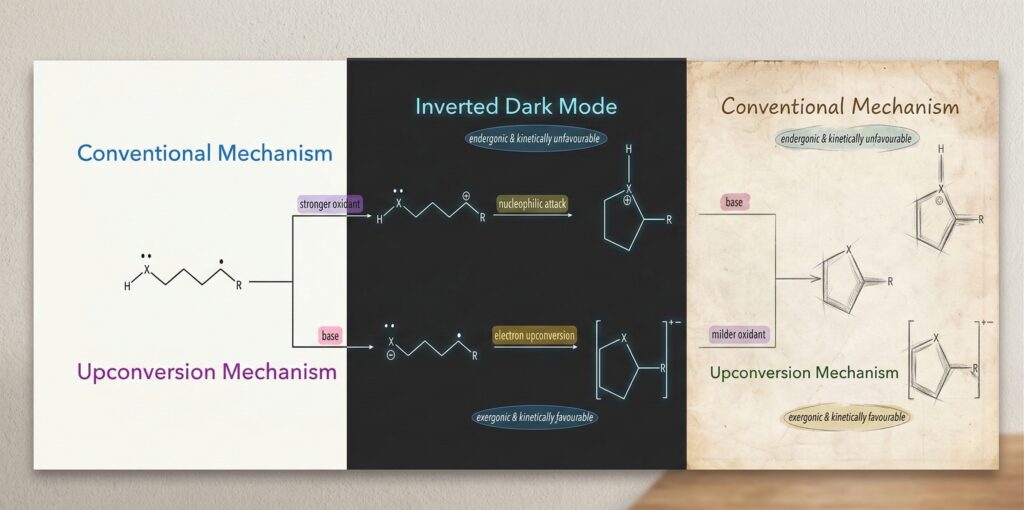
Electron Upconversion Enables CP and CS Bond Formation Under Mild Oxidative Conditions: A Theoretical Study
Meera's first paper as the part of her PhD thesis published in Journal of Computational Chemistry. The work has been based on some of the finidngs of Prog Igor Alabugin of Florida State University, USA. Traditional methods for forming carbon–phosphorus (C–P) and carbon–sulfur (C–S) bonds often rely on strong oxidants that generate high-energy carbocation intermediates, frequently leading to unwanted side reactions. In this work, we employ DFT to investigate an alternative mechanism operating under basic conditions that utilizes three-electron bond formation and concomitant electron upconversion. Our computational results reveal a radical-anionic pathway initiated by a cyclization to form a 2-center-3-electron (2c-3e) bond, followed by the oxidation of the resulting upconverted radical-anion by mild oxidants such as molecular oxygen . This pathway is shown to be both thermodynamically and kinetically favored over conventional two-electron routes, providing a more controlled and selective strategy for C–P and C–S bond construction. These findings suggest that electron-upconversion mechanisms can significantly advance green chemistry by reducing dependence on harsh reagents and minimizing synthetic side reactions.
View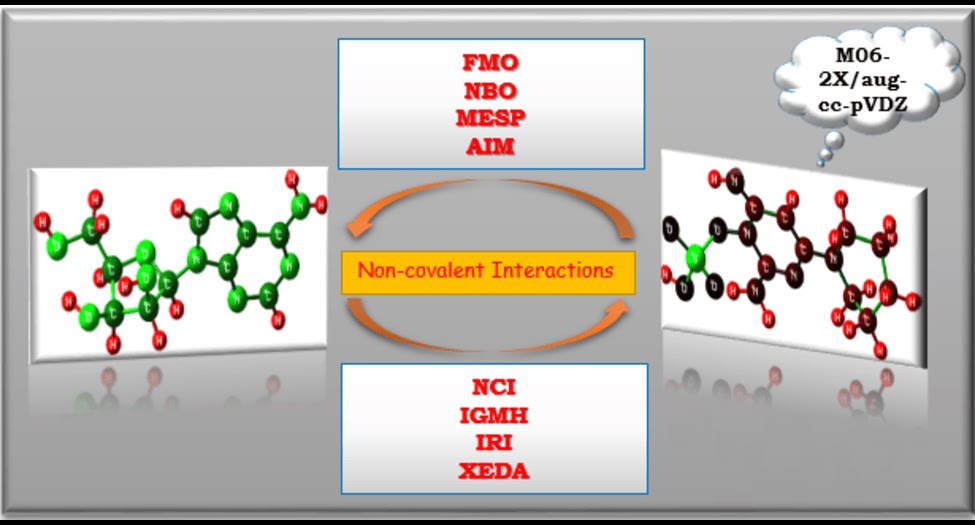
Sneha & Ananya’s paper in Journal of Physical Chemistry B
PhD student Sneha Anna Sunny's paper on the Mapping the Interaction Landscape of Adenosine and Minoxidil Sulfate Using an Independent Gradient Model Based on Hirshfeld Partition and Interaction Region Indicator. Ananya Prakash is a final year MSc Physics student of our college who worked in our lab as part of the Kerala Theoretical Physics Initiative (KTPI) student project initiative,
View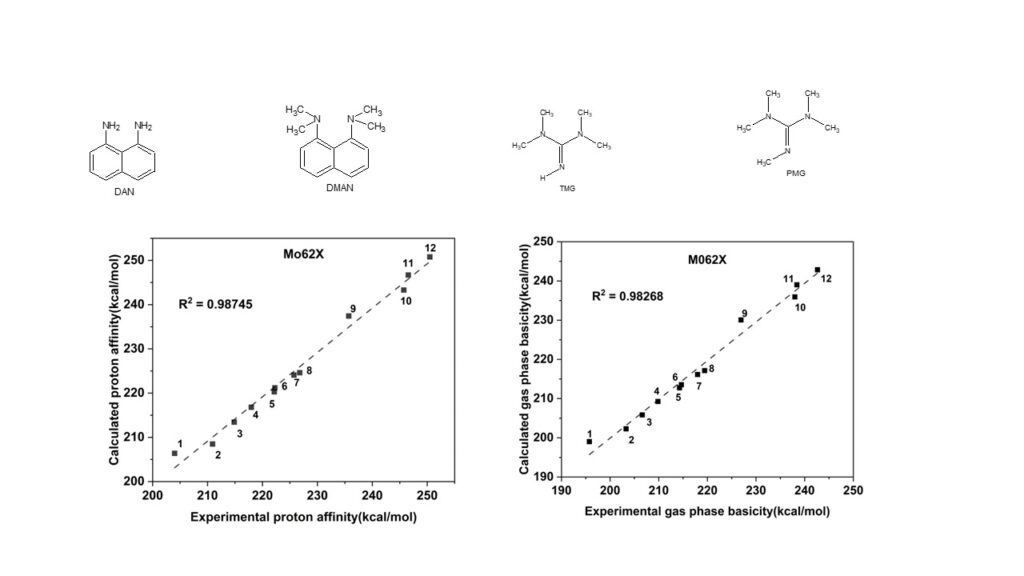
Manjesh Mathew: Benchmarking Computational Approaches for Proton Affinity
In this collaborative study with Dr Ralph Puchta, Manjesh Mathew, PhD student assessed the proton affinities and gas phase basicities of molecules ranging from ammonia to proton sponges like PMG using computational methods including B3LYP, BP86, PBEPBE, APFD, wB97XD, and M062X with the def2tzvp basis set. The M062X method showed the highest accuracy, closely matching experimental results for all examples, especially those containing heteroatoms, while APFD and wB97XD tended to overestimate values. Dispersion corrections were evaluated but found not to significantly affect basicity predictions. Computational and Theoretical Chemistry
View
Alen Binu Abraham : Non-Covalent Interactions of Caffeine and Ascorbic Acid
In this study, we explored the non-covalent interactions between caffeine and ascorbic acid, focusing on their hydrogen bonding in both gas and solvent (water) phases, using Density Functional Theory. The binding energy calculations revealed significant interactions with values of −14.65 kcal/mol in the gas phase and −11.62 kcal/mol in water. Through Natural Bond Orbital, RDG, AIM, and LED analyses, we confirmed the stabilization energy and electron delocalization in the caffeine-ascorbic acid complex, enhancing our understanding of drug-drug interactions and their implications for drug efficacy and delivery systems. Abraham, Alen Binu, Alzahrani, Abdullah Y. and Thomas, Renjith. "Exploring non-covalent interactions between caffeine and ascorbic acid: their significance in the physical chemistry of drug efficacy" Zeitschrift für Physikalische Chemie, vol. 238, no. 2, 2024, pp. 401-420.
View
Gayathri: Hydrogen Bonding and Dynamics of Prontosil in Water and Methanol
In this theoretical study, we investigated the solvation energies of prontosil, a pioneering sulfa drug, in water and methanol using DFT/M06-2X/cc-pVDZ level of theory. Our findings reveal that prontosil shows more favorable interactions with methanol than water, as evidenced by NBO, RDG, QTAIM analyses, and local energy decomposition (LED) analysis using DLPNO-CCSD(T). The prontosil-methanol complex displayed the lowest binding energy and highest stability, suggesting methanol as a more suitable solvent for prontosil in terms of molecular interactions and stability. These insights are further corroborated by ab initio molecular dynamics simulations, underscoring the importance of solvent choice in drug design and molecular interaction studies. Here is the link to "Understanding the hydrogen bonding preferences and dynamics of prontosil in water and methanol":
View
Jisha’s paper in Journal of Physical Chemistry-B (ACS)
Postdoc Dr Jisha's latest paper Assessing the Noncovalent Interaction of Deucravacitinib and Ethanol with Special Reference to an Independent Gradient Model Based on Hirshfeld Partition
View
Rajimon’s first bioactive chalcone paper in JMS
Rajimons Chalcone paper in collaboration with Prof Sarojini, Mangalore Univeristy
View
Metalfree cycloaddition in New Journal of Chemistry
The paper investigates the metal-free [3+2] cycloaddition reaction between phenyl azide and phenyl enaminone to selectively synthesize 1,2,3-triazoles. Using computational methods, it explores the reaction pathways (1,4- and 1,5-pathway), solvent effects, and the impact of phenyl azide substituents, offering insights into the reaction mechanism and the role of water as a solvent and catalyst.
View
Rajimon : Schiff base derived from 4-Chloroaniline and 2-Formylphenol
PhD student Rajimon KJ's 6th first-authored paper in Journal of Molecular Structure (Elsevier). Comprehensive assessment of schiff base derived from 4-Chloroaniline and 2-Formylphenol: Molecular architecture, experimental with computational bioactivity profiling, emphasizing anticancer efficacy against pulmonary and mammary carcinoma cell models
View
Cu-Schiff base complex in Polyhedron
PhD student Rajimon KJ's 7th first-authored paper was published in Polyhedron (Elsevier). Structural, spectroscopic, and biological properties of an asymmetric Cu (II) Schiff-base complex: Synthesis, DNA/BSA interactions, and antimicrobial potential with experimental and computational insights
View
Collaborative paper with Prof BK Sarojini, Mangalaore University
Supramolecular and computational analysis of Fluconazole −2−chloro−5−nitrobenzoic acid cocrystal. PhD student, Rajimon KJ was a visitor at her lab last year.
View